Congratulations to Professor Yoshinori Ohsumi Tokyo Institute of Technology on being awarded the 2016 Nobel Prize in Physiology or Medicine for “for his discoveries of mechanisms for autophagy“!

The process of autophagy is hardly one familiar to most people, but is is absolutely crucial to all complex life on out planet, including ourselves. The name autophagy comes from the Greek words for “self” and “eating” and describes the ordered process through which cells break down and recycle unnecessary or damaged structures or proteins, and allows the cell to reach an equilibrium between the synthesis and degradation of proteins.
The discovery of autophagy
The process itself was identified through studies in tissues of mice and rats back in the 1950’s and 1960’s, by scientists including Christian de Duve, who was subsequently awarded the Nobel Prize in Physiology or Medicine in 1974 for this and other work. They first discovered that mammalian cells contain a compartment which they termed the lysosome where proteins are broken down, and then that proteins and other molecules that were to be degraded were first isolated from the rest of the cell by the formation of a membrane sac around the protein in question (later called the autophagosome). The process through which the autophagosome fused with the lysosome to deliver its protein cargo for degradation was given the name autophagy by Christian de Duve.
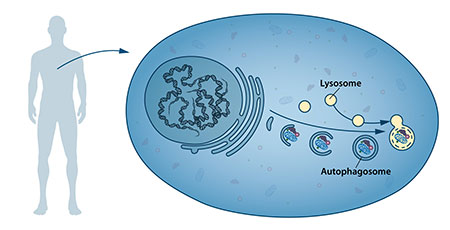
Progress in understanding how autophagy worked was slow, as at the time the genes or proteins involved in regulating the process had been identified. With the research methods available at the time it was difficult to measure autophagy as it happened in mammalian cells, and hence difficult to determine how altering different components affected the overall process, a key step towards understanding their role. It may have seemed an unpromising field to join, but Yoshinori Ohsumi had a different career philosophy to most researchers, which he described in an interview given in 2012:
I am not very competitive, so I always look for a new subject to study, even if it is not so popular. If you start from some sort of basic, new observation, you will have plenty to work on.
From cells to genes
What was needed was a simple experimental system in which to study the process, and the bakers yeast Saccharomyces cerevisiae – a simple single celled organism separated from us by hundreds of millions of years of evolution, but sharing many of our key biological processes – was one candidate. Yoshinori Ohsumi had worked with yeast, and in particular had identified many proteins in a subcellular component of the yeast cell known as the vacuole, which was important as there was evidence that the vacuole performed the same role in yeast cells as the lysosome in mammalian cells. Still, as the Nobel Prize website highlights there were still hurdles to overcome as he began his study of autophagy in yeast at the end of the 1980’s:
But Ohsumi faced a major challenge; yeast cells are small and their inner structures are not easily distinguished under the microscope and thus he was uncertain whether autophagy even existed in this organism. Ohsumi reasoned that if he could disrupt the degradation process in the vacuole while the process of autophagy was active, then autophagosomes should accumulate within the vacuole and become visible under the microscope. He therefore cultured mutated yeast lacking vacuolar degradation enzymes and simultaneously stimulated autophagy by starving the cells. The results were striking! Within hours, the vacuoles were filled with small vesicles that had not been degraded (Figure 2). The vesicles were autophagosomes and Ohsumi’s experiment proved that authophagy exists in yeast cells. But even more importantly, he now had a method to identify and characterize key genes involved this process.
With an experimental system available Yoshinori Ohsumi and his team studied the process of autophagy in thousands of mutant strains of yeast, and identified 15 individual genes (most of them of previously unknown function) that are essential for the process in yeast, tho order in which the key events in autophagy take place, and the roles of the individual genes in them. This was the work for which he was awarded the Nobel Prize.
From yeast genes to us!
But it is not the end of the story! Identifying the genes essential for autophagy in yeast, and their roles in the process, was a major breakthrough, but what about humans and other mammals?
It turns out that that in humans and other mammals there are counterparts to almost all the yeast autophagy genes, though the situation is made a lot more complicated by the face that mammals have more than one copy for each of the genes…starting with yeast was a wise move! Professor Noboru Mizushima of the University of Tokyo made an important advance when, working with Yoshinori Ohsumi, he developed a transgenic mouse in which a protein called LC3 that is found in the autophagosome membrane is fused to Green Fluorescent Protein (GFP – see Nobel Prize for Chemistry 2008) which allowed him and his colleagues to observe and monitor the process of autophage in vivo in mice for the first time.

This LC3-GFP transgenic mouse proved to be a very powerful research tool for studying mammalian autophagy, allowing not only the role of indicudual genes in the process to be determined, but also the role of autophagy itself in processes as diverse as early embryonic development, tumor suppression, nerve cell survival and function, and protection against infection.
This research is still at a relatively early stage, but techniques such as the LC3-GFP system in mice – and others used in organisms such as fruit flies, are showing us how defects in autophagy contribute to many diseases, including neurodegenerative disorders such as Parkinson’s Disease, and metabolic disorders such as type 2 Diabetes. While the development of specific therapies to correct these defects in autophagy is still some way off, it is already clear that understanding autophagy has the potential to improve the treatment of a wide range of illnesses.
What the work of Yoshinori Ohsumi demonstrates once again is the crucial contribution of basic biological research in model organisms that may at first glance appear to share little with us to the advancement of medicine.
Speaking of Research
