Unfortunately, the “how” and “why” of the research process is of much less interest, and receives far less attention, than the “what did they find?!” part of research. The latter is what you’ll see—if we’re lucky from the science outreach perspective— on television, in the science and popular media, Facebook, Twitter, and conversations world-wide. Meanwhile, the former will be relegated to websites of federal agencies, scientific societies, and animal research advocacy groups and are read less widely. In fact, it is entirely possible that a great many bets could be won by wagering that the public generally doesn’t care to read up on regulation or processes governing the research behind the cool discoveries that make news.
In the case of animal-based research (and some other controversial fields), the “how” and “why” do sometimes generate some public interest because they are keystones in considering questions about its ethical basis and evaluation. Public understanding and discussion of the process by which science moves forward is important. It provides appropriate context for fact-based dialogue about the ethical evaluation, decision-making, and regulation that govern a wide range of science conducted within our democratic system. Thus, many scientists and advocates not only welcome public interest in the conduct of science, but also actively promote thoughtful, engaged, and informed collaboration on efforts for improving research practices.
Why? One reason is that the ultimate benefactor from scientific studies is the public and, within a democratic society, it is for all of us to decide whether the benefits of those studies outweigh their costs. Another reason is that scientists are generally sensitive and responsive to societal views, but feel an obligation to ensuring that these views are informed by facts as well as emotional appeals. This is an issue that is not at all unique to animal research. It also appears in discussions of other topics that can elicit controversy, including for example: evolution, climate change, use of embryonic stem cells, and vaccines.
For animal research, the challenges inherent in serious evaluation of its costs and benefits are not trivial. Nor is it amendable to flashy, sensationalized, and emotion-evoking campaigns. Simplistic approaches to this issue are not useful and do a disservice to all of us.
From our perspective, it is both disappointing and frustrating to find that understanding of the process by which science moves from idea, to the conducting of the study, to the dissemination of the findings, to the evaluation of those findings receives far less attention than would be needed in order to rationally discuss the research. Why? Because the reality of how science is actually conducted is centrally relevant to conversations about science. And while this is an obvious statement, it is also clear from many portrayals of science by opposing groups that the basics of scientific process and conduct are often missed in the discussion.
In the case of laboratory animal research, the starting point of many opponents is an absolutist position in which the conditions for animals, the ultimate outcome of the research, and its benefits, are irrelevant. They are irrelevant because the starting assumption is that the use of animals is morally unacceptable. For those who hold this view, there is no benefit that would justify the animal use. There are others who hold a less absolute view and, like us, believe that the use of animals in research begins with moral and ethical consideration that requires thoughtful, fact-based weighing of both relative harm and benefit. One major part of this evaluation is identifying whether alternatives exist to meet the same goal. Another is identifying as closely as possible what harm may be incurred, the probability and extent of benefits. Each of these considerations is integral to regulation of animal research in the U.S. and elsewhere. They are also considerations that are so integral to the scientific process that they operate far beyond those stages typically identified as the “checks” for ethical and humane conduct of animal research (e.g., IACUC review, federal oversight).
How scientific research moves from idea stage, to conducting a study, to success or failure, to critical review, to dissemination and use of findings is a process that can appear somewhat opaque to public view. The pieces of information required to construct the general pathways are publicly available. Putting them together, however, is not necessarily straightforward for those without immediate interest, expertise, or engagement. So while the information is neither hidden nor made secret, it is of the type that can be easily misunderstood or misrepresented.
Should this gap in basic understanding and perspectives on how scientists’ ideas move from thinking to reality concern us? The answer is yes. Among other reasons, the gap serves as an impediment to an informed evaluation of science. It also weighs heavily against productive dialogue about core issues of public interest.
How does an animal research project move from scientist’s idea to finished study?
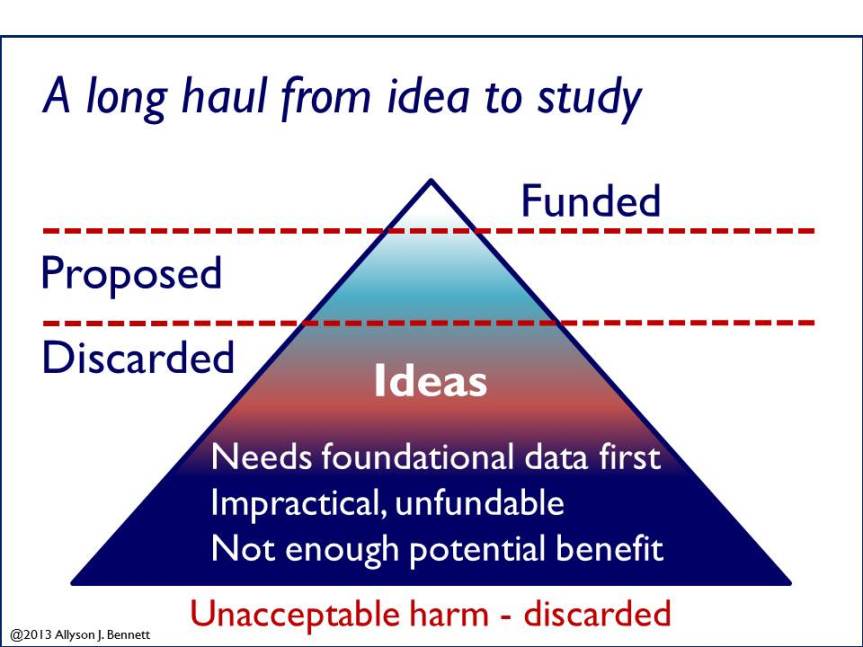
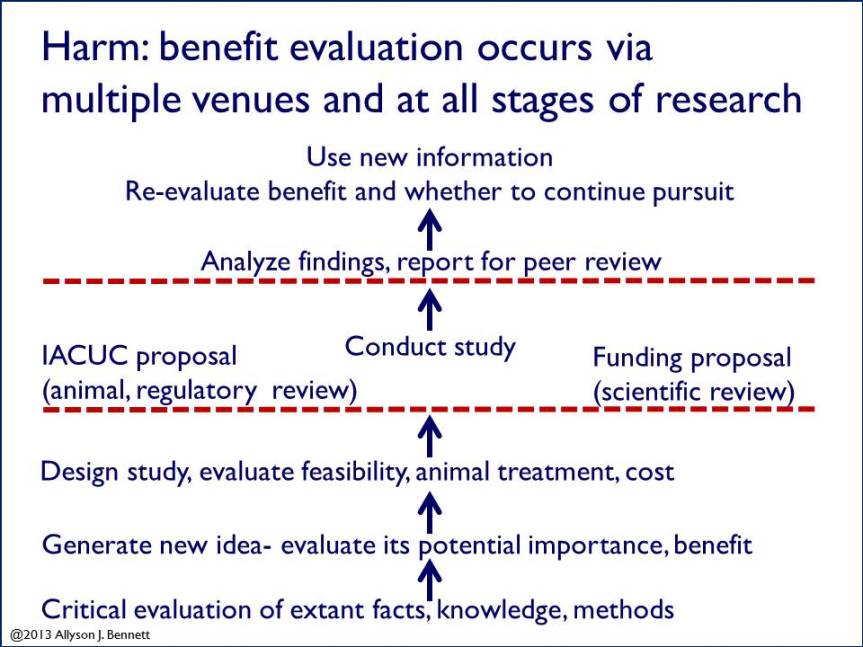
In general, the process looks like this: Scientists generate ideas that are based in careful study of what is known, what is not known, what methods already exist, what facts we have. They next critically evaluate and review relevant previous literature and data– often soliciting others’ expert knowledge– to determine whether the idea is novel (has not already been tested), of potential importance or significance, and feasible.
Thus, while some may have the impression that scientists roll out of bed in the morning, or have an aha-moment- then move straight to the lab to conduct whatever study occurred to them via dream – this is not the way it typically works.
As illustrated, deciding on whether an idea is worth pursuing or not is driven by many factors. If the resulting data would have little potential benefit, few scientists are likely to pursue it. Why? Because scientists have a lot of ideas and it makes no sense to expend energy on one that won’t be useful in terms of providing significant new knowledge or understanding. It is also true that such ideas are unlikely to compete successfully in the different arenas of expert scientific review, including review for funding, publication, and citation.
If a scientist judges his/her idea worth pursuing, the next step is likely to decide whether the study is feasible or practical. What does this mean? In short, this is a question that revolves around ethical, economic, and practical issues. On the ethical side, for animal research the scientist will consider animal welfare and treatment, any potential for harm. Next, on the financial and practical sides, the scientist will consider how much the study will cost and whether the necessary work can even be done. During this initial stage the scientist will also critically evaluate whether the existing literature and facts provide adequate and strong platforms for the proposed study, or whether more basic and background data are needed to guide decisions before moving forward.
For that fraction of studies that survive the scientist’s own critical examination—and likely that of his/her collaborative group and colleagues—the scientist may decide to pursue the work. If so, for animal research the next step will be to write a proposal to the Institutional Animal Care and Use Committee (IACUC) in order to conduct a study. In the U.S., IACUSs are among the main venues for thorough review of animal studies. We have written previously about IACUCs and there is more information here.
In brief, the IACUC is comprised of individuals with veterinary and scientific expertise, as well as a public representative. Animal studies do not proceed until the IACUC has reviewed and approved a proposal. What do these protocols contain? You can see some here, this site contains links to protocol forms from a range of institutions. Although institutions vary in the format of applications, among other things, they include: information about what the study is designed to test, why it should be conducted, the literature review and strategies used to ensure that it is not unnecessarily duplicative, that alternatives do not exist, the number of animals proposed and justification for both the number and the species, detailed description of all procedures, and other details about the animals’ care and treatment. In other words, the full range of information that the review committee will need in order to evaluate whether the study meets standards.
Is the IACUC process perfect in evaluating study protocols? No. It is, however, the current system mandated by federal law and it is one that generally functions well to protect animal welfare. It is also an evolving system, with scientists, veterinarians, federal agencies, science and animal welfare advocates engaged in its ongoing evaluation and improvement. Some of the criticisms of the existing system, however, neglect consideration of the larger context, the process by which research unfolds. For example, critics point to the fact that IACUCs approve the majority of studies put before them as evidence that “almost anything” a scientist could dream up receives approval. In reality, IACUCs only review proposals that scientists write and submit. This means that the IACUC only sees study proposals that have already received some critical evaluation and that likely already fall within the constraints of current guidelines, practices, and norms. Scientists, like others involved in animal research, take part in training and education about the range of issues related to animal welfare, humane treatment, and regulatory requirements. As a result, they are generally not likely to write protocols that diverge from acceptable practices.
Following IACUC approval, the scientist may then begin conducting the study. It is often the case however, that IACUC approval is not the final step between idea and study. Instead, for a new project, the scientist must also write a proposal to a funding agency in order to secure financial support for the research. In many cases in academic research, funding for these studies comes from federal agencies such as the National Institutes of Health or the National Science Foundation. Competition for these funds is high and the majority of applications are not successful. Those proposals that are funded have undergone rigorous review by a panel of scientists whose expertise is within the area of the proposal. The criteria for review vary by agencies, but include very close examination of the significance of the research question, evaluation of its potential for success, scrutiny of the methods, expertise of the investigator, and quality of the facilities in which the research will be conducted. The appropriateness of the animals chosen for study, their number, and their treatment are also subject to critical evaluation and discussion. In sum, beyond IACUC review, many animal studies—including all of those funded by NIH, NSF, and other agencies— undergo another level of external expert scientific review.
Take-home message? The evaluative process between a scientific idea, the conduct of a study, the results, and their evaluation, use, and further discovery is one with many steps and significant consideration. The potential harm and benefit of each study receives review at each stage as well, both within and outside.
Research aimed at addressing basic, translational, or clinical questions relevant to advancing our scientific understanding and medical progress for humans and other animals is ultimately all aimed at questions with significance to many. At the same time, it is also absolutely true that the benefits of research are not always directly or immediately apparent. We simply do not know the answers before we conduct the work. Furthermore, we can be confident—drawing from real conclusions from the history of science – that important, meaningful, generative breakthroughs are not entirely predictable. As a result, it is no easy task to construct a metric by which to evaluate the potential benefit of research and to weigh that against any harm incurred during its conduct.
Considered carefully, the history of animal research and animal welfare are quite clear with respect to how the accomplishments of research and consideration of mutual interests in animal welfare provide the basis for progress in ethical and humanely-conducted animal research. Public interests are served by dialogue based in fact and in clear accurate articulation of ethical frameworks from which animal research is considered. Understanding the multiple levels at which research projects are evaluated from scientific and ethical perspectives is an integral starting point for this discussion. Science doesn’t occur through simple processes or via a single stage of evaluation; nor should public dialogue about this complex issue.
Allyson J. Bennett

Reblogged this on disce aut discede!.
Yes, these dogs are clearly anesthetized and are in temporary holding (note: there are no food & water dishes on the doors). Have you ever been to a veterinary clinic? One difference is these cages are much cleaner. The techs seem very gentle with them and are talking nicely to them. Yes they are on a study, but seem to be very well cared for.
How did you relieve their suffering Tom?
In the video you have several dogs which look very clearly anaesthetised.
I can’t say I’m too impressed with the cages. I don’t know when this video was taken but there is a strong movement towards larger social housing for dogs. Also with more enrichment. I don’t know if these are temporary cages for dogs on the study or if it’s a lab which hasn’t made the transition to better housing (also depends how long ago this was taken).
I’m guessing this is toxicological research (which is mandatory for new medicines). The point is to discover whether something is safe for early clinical trials – all you can do is provide pain relief.
Note the dogs being transported through the corridors (who are probably not on a study) look a lot happier – animal technicians do their best to enrich the lives of the animals.You will also note the inections at the end are carried out much as they would be at a veterinary practise.
Since “Tom” likes to say about how they don’t make animals suffer and use “anesthesia” what about the psychological torture that is deliberately done for research purposes? How do you alleviate that? What about exploding hearts that are deliberately made that way, how do you alleviate that suffering? What about all of the repulsive addiction research? What has that brought any of us, and how do you alleviate that suffering.
What if I told you that I have a disease that has no cure, and that if one is ever created using animal testing, I will refuse it.
What’s with the speech marks? Are you questioning my name being Tom or the existence of anaesthetic?
What psychological torture are you on about? Provide a link to some research in the past 20 years and we can try and give you an answer.
If an animal is going to have an induced heart attack it will be anaesthetised. That’s how you alleviate suffering.
Addiction research is ongoing. Scientists understand a lot more about the neural and chemical pathways of addiction, and hopefully this will lead to a cure – this sort of stuff doesn’t happen overnight.
If you said you have a disease which you would not take medicine for then you are more principled than more activists. I suppose you never take medicines then? (Though even if you didn’t you would still benefit from the vaccinations of others).
Sorry, to be more clear, I had seen what I thought was a comment or a blog entry about how the reactions of people to experimentation was different because of some psychological/psychosomatic reaction that it made testing on people unhelpful scientifically.
Didn’t you have a F&Q page or a blog entry that refuted the argument for replacing animal testing for testing on pedophiles? I’ve been looking high and low for it, but can’t find it.
If it doesn’t exist, can you tell me why experimenting on death row inmates, pedophiles and all the other dregs of society instead of animals is not a good idea?
We’ll consider a blog post on that – good thought
In the meantime, the quick answer is that the Nuremberg Code, developed after the Nuremberg Trials post WW2, states that experiments using human subjects “should be so designed and based on the results of animal experimentation….such that the anticipated results will justify the performance of the experiment.” (http://en.wikipedia.org/wiki/Nuremberg_Code). There are nine other tenants of the code which all boil down to you can’t experiment on humans without their consent.
“In sum, beyond IACUC review, many animal studies—including all of those funded by NIH, NSF, and other agencies— undergo another level of external expert scientific review.”
I’d add that most charitable foundations also require grant proposals to be reviewed by expert panels before selecting the proposals they will fund. These charities organizations such as the Gates foundation, American Cancer Society, and Howard Hughes Medical Institute – and many more smaller foundations – that between them invest several $billion in medical research every year.