Welcome to this week’s Research Roundup. These Friday posts aim to inform our readers about the many stories that relate to animal research each week. Do you have an animal research story we should include in next week’s Research Roundup? You can send it to us via our Facebook page or through the contact form on the website.
- Brain cells from Pigs have been implanted into Parkinson’s patients in hopes to stop the progression of the disease. Parkinson’s is a neurological disease in humans that causes tremors and difficulty moving, which usually worsens over time.
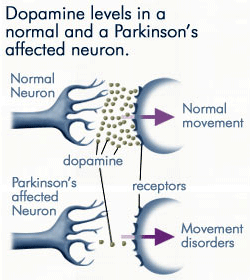 Such dysfunction of the motor system is caused by gradual loss of brain cells that make dopamine. Current technologies synthetically replace dopamine in the brain, however, this is not always effective. Now, a new technology, developed by Living Cell Technologies, implants cells from the choroid plexus of pigs into humans with the aim of nourishing the remaining dopamine-producing cells. The approach was first successful in a rat model and non-human primate model of Parkinson’s disease and has now been done in 4 human Parkinson patients with promising results 18 months after implantation.
Such dysfunction of the motor system is caused by gradual loss of brain cells that make dopamine. Current technologies synthetically replace dopamine in the brain, however, this is not always effective. Now, a new technology, developed by Living Cell Technologies, implants cells from the choroid plexus of pigs into humans with the aim of nourishing the remaining dopamine-producing cells. The approach was first successful in a rat model and non-human primate model of Parkinson’s disease and has now been done in 4 human Parkinson patients with promising results 18 months after implantation.
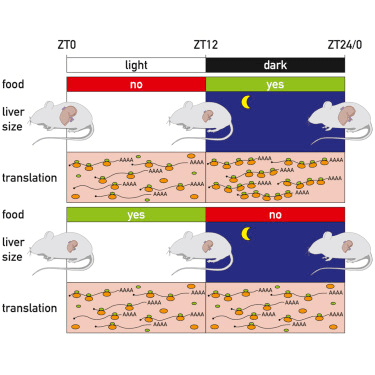
- A new study suggests that the liver may grow and shrink by up to 40% every 24 hours in response to ribosomal activity. Mammals, in general, consume food at certain times throughout the day (breakfast, lunch, and dinner), in a cyclical rhythm. After consuming this food, the liver plays a pivotal role in producing biochemicals necessary for digestion, regulating glycogen storage, and detoxifying byproducts of metabolism. Recent research, published in the journal Cell, investigated how the liver adapts to daily rhythms of eating using mice as a model of mammals. Their research showed that the mass of the liver, hepatocyte size, and protein levels follow a daily rhythm, that depends on feeding-fasting and light-dark cycles. A second experiment showed that daily rhythms of protein levels in the liver are correlated with daily rhythms in ribosome number. This fundamental research has implications for our general understanding of liver function, which may allow for future cures in liver disease and dysfunction.
- Lab-grown cartilage has similar mechanical and biochemical properties to natural cartilage. Cartilage helps joints to move, but can easily be damaged by trauma, disease, or overuse. Once damaged, cartilage does not regrow and is often difficult to replace. Biomedical engineers have been developing artificial cartilage using human chondrocytes over the past several years to replace damaged cartilage. Now, researchers from the University of California Davis have tried a new method by growing the artificial cartilage under tension, which helped the cartilage grow stronger and now has similar properties of natural cartilage. The researchers then implanted cartilage into mice and found there were no negative interactions between the cartilage and the living mouse. The next step is to try the lab-grown cartilage on a load-bearing joint to see if it remains durable under stress.
- Social stimuli may be an inadequate replacement for juice rewards for monkeys in behavioural neuroscience research. A study funded by the NC3Rs aimed to find out if images of other monkeys could be used to reward monkeys for participating in research, rather than traditional juice rewards — which often require fluid-restrictions to work. The researchers conducted the study in 4 rhesus macaques, and first confirmed that these monkeys preferred monkey images (a social stimuli) to nonsense control images. They then tested monkeys on a simple cognitive task, offering only juice reward, juice + social stimuli, or only social stimuli. In all monkeys the juice reward improved motivation, and only one monkey did the social stimuli improve motivation. The scientists concluded that this form of social stimuli might be ineffective, suggesting it may be because all monkeys are pair housed in socially stimulating environments. The study was published in PLOSone.

- 8 years of research culminate in a vaccination to fight heroin addiction. Approximately 9.2 million people in the world use heroin, some of which results in death due to heroin abuse — approximately 91 Americans die each day from overdosing on opioids. Using mice and rhesus macaque monkeys, researchers at the Scripps Research Institute developed a heroin conjugate vaccine which reduced the potency of heroin by 15 times in mice and 4 times in monkeys. The effects of these vaccinations persisted for over 8 months. This preclinical research brings us closer to an effective heroin vaccination for treating opioid use disorders. This study was published in the Journal of the American Chemical Society.
