Welcome to this week’s Research Roundup. These Friday posts aim to inform our readers about the many stories that relate to animal research each week. Do you have an animal research story we should include in next week’s Research Roundup? You can send it to us via our Facebook page or through the contact form on the website.
- Universal flu vaccine shows promise. Scientists have successfully tested a promising vaccine that protects against many types of influenza for over 30 weeks. Current flu vaccines target unique surface proteins of influenza called hemagglutinin — particularly, the heads. These hemagglutinin heads rapidly mutate making it difficult to protect against different types of influenza and requiring annual immunizations. The new study targeted the stalks of the hemagglutinin, rather than the heads, which do not vary between different influenza subtypes. Current research tested the vaccine on mice, ferrets, and rabbits. The vaccine should reach clinical trials within the next two years, and the market in ten. Published in Nature Communications.
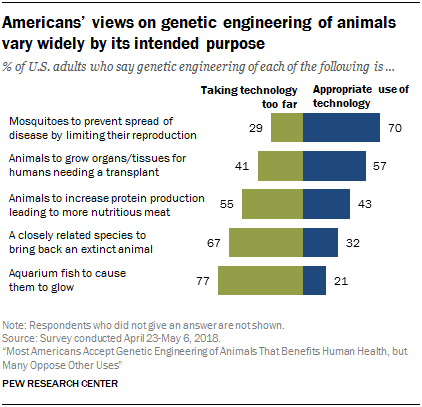
- New poll data shows support for genetic engineering of animals that benefits human health. Genetic engineering of animals is an emotionally fraught topic–particularly when considering that genetic engineering serves multiple purposes. When considering the intended use of these technologies, majority support was found only for those that have the capacity to pre-empt or ameliorate human illness; animal research continues to play an integral role in this process.
- New anti-obesity drug discovered while researching insulin resistance. While studying a drug that prevents insulin resistance scientists discovered that it actually prevented mice from depositing and storing fat. The current study designed a drug to reduce the risk of insulin resistance by inhibiting the enzyme ceramide synthase 1 (CerS1). When testing the drug in mice however, the drug increased their ability to burn fat in skeletal muscles and had no direct effect on insulin resistance. Although the study did not accomplish its main objective, it may lead to future anti-obesity drugs. It is also the first drug to successfully target enzymes in the ceramide family, which are associated insulin resistance. Published in Nature Communications.
- Research in mice has helped solve a two-decade-old metabolism mystery. Scientists at the University of Michigan and Vanderbilt University have determined the function of the melanocortin 3 receptor (MC3R). According to researchers, the cell receptor is involved in “energy rheostasis,” which controls weight loss and weight gain when an organism’s metabolism is challenged. Mice lacking this protein tend to lose more weight when ingesting fewer calories and gain more weight when eating higher-fat diets. MC3R’s function makes it a promising new drug target for treating obesity. Published in Science Advances.
- New gene therapy saves night vision in canine eye disorder. Retinitis pigmentosa is a group of hereditary eye disorders in which people lose night and peripheral vision before losing sight altogether. Researchers at the University of Pennsylvania developed the first gene therapy that prevents the loss of night vision in a canine-version of the disorder. With this therapy, both the abnormal and normal copies of the gene responsible for the disorder, known as rhodopsin, are disabled and a healthy copy is provided. Approximately 30% of normal levels of healthy rhodopsin was achieved with treatment, which enabled the transmission of light signals to the brain and preserved night vision in six dogs. These results will pave the way for clinical trials in humans. Published in the Proceedings of the National Academy of Sciences.

