June 3rd 2022
The search for a fountain of youth has haunted humans since the beginning of time. Now, researchers from Stanford University reported in the peer-reviewed journal, Nature1, that older mice receiving cerebral spinal fluid (CSF) from younger mice had their memory impairment reversed. Through a series of rigorous experiments spanning behavioral, cellular, and molecular analyses, the team identified a substance contained in CSF, a fibroblast growth factor called Fgf17, as responsible for the remarkable memory improvement. Specifically, when aged mice were given Fgf17 in a brain structure called the hippocampus, the substance promoted proliferation and differentiation of cells called oligodendrocyte progenitor cells (OPCs). OPCs are precursors to oligodendrocytes, cells which help support the wiring of the brain. The study suggests that restoration of oligodendrocyte function and myelination in the aging brain may help prevent the cognitive decline associated with aging.

The study involved 20-month-old mice, half infused with artificial CSF (control group) and half infused with CSF from young mice. For CSF infusion, a tiny hole was drilled in the skulls by a skilled surgeon and a pump was implanted on their upper backs. The mice were trained on a fear conditioning task, a primary screening tool used in biomedical research for investigating deficits in memory. In this task, mice received 3 (mild, yet aversive) foot shocks associated with a cue (tone and flashing light). Three weeks later, they were re-exposed to the cues (tone and light) to see how well they remembered the foot shock associated with the cue. The old mice infused with young CSF had better recall than the control group that did not receive Fgf17.
Because learning and memory are dependent on the hippocampus, the researchers studied the hippocampus next. They performed RNA sequencing to identify genes differentially expressed in the two groups of mice. They found that oligodendrocyte genes were highly increased in the group receiving CSF from young mice. Consequently, they wondered whether this could be due to OPC proliferation and differentiation in the hippocampus.
To test this hypothesis, they labeled dividing cells in the last 2 days of young CSF infusion and observed a surge in cell proliferation specifically in the hippocampus. To see whether young CSF could promote OPC proliferation and differentiation directly, they turned to OPC cell cultures. They found out that the top gene induced after 1 h of infusion with young CSF was for the serum response factor (SRF). Through a series of experiments, they discovered that SRF signaling in OPCs was decreased with aging and increased by acute injection of young CSF in vivo.
Many proteins in CSF could have induced SRF signaling, and one of the problems was to zero in on one particular substance. They identified Fgf17, a fibroblast growth factor, as a potential candidate. Sure enough, FgF17 induced OPC proliferation in the aged hippocampus and improved long-term memory performance in aged mice. Conversely, blocking FgF17 in young mice impaired memory performance and neuronal plasticity, and prevented OPC proliferation induced by young CSF or FgF17 in cell cultures (Fig 2).
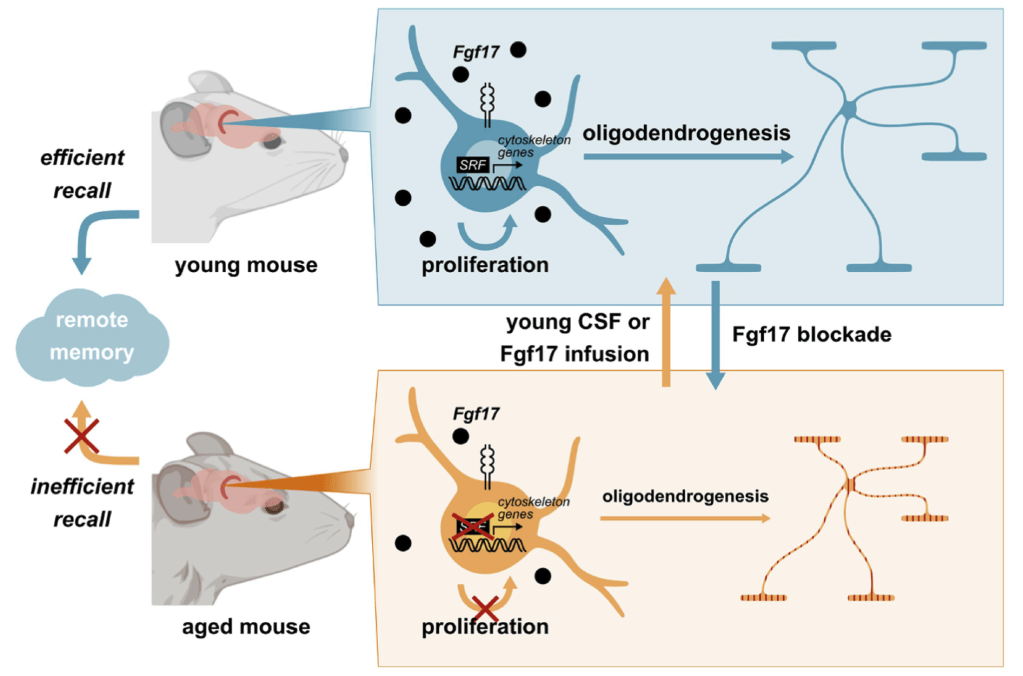
The results of these experiments demonstrate that Fgf17 is a major component of the rejuvenating effects of young CSF in mice. They also suggest that targeting hippocampal myelination might be a therapeutic strategy to prevent or rescue age-related cognitive decline. Of course, it remains to be seen whether targeting hippocampal myelination will also be an effective strategy against age-related cognitive decline in other animals beyond mice, like primates. As Fgf17 also decreases with age in human CSF and plasma, it is worth finding out.
~Speaking of Research
1Iram, T., Kern, F., Kaur, A., Myneni, S., Morningstar, A. R., Shin, H., . . . Wyss-Coray, T. (2022). Young CSF restores oligodendrogenesis and memory in aged mice via Fgf17. Nature, 605(7910), 509-515. doi:10.1038/s41586-022-04722-0
