June 30th 2021
The United Kingdom reports a wealth of information every year on its use of animals in research. Every animal procedure must be categorized according to the overall purpose (and specific disease area or regulatory purpose if relevant), the genetic status of the animals and the severity of the procedure. Historically, Great Britain (England, Scotland and Wales) conducts approximately 99.5% of procedures, which are regulated by and reported by the UK Home Office. Northern Ireland has not yet released animal use statistics for 2021.
Animal Research in the United Kingdom (excluding Northern Ireland)
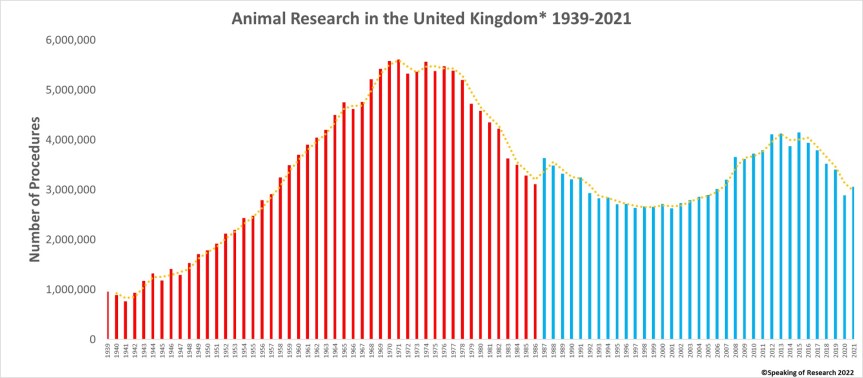
Every year the Home Office releases the statistics on animal research in Great Britain (Northern Ireland released separately) for the preceding year. In 2021, the total number of procedures on animals was 3.06 million, an increase of 6% from the 2.88 million in 2020.
The total number of animals used is quite likely lower as some animals were used for more than one procedure*. In 1987 the UK changed the way in which it counted animals, from the total number of animals to the total number of procedures. There was a further, smaller change to methodologies in 2014.
*The total number of animals excludes those animals first used in a previous year, which underwent a procedure in the current year, in order to prevent double counting of animals between years.
The last few years have seen the number of Genetically Altered animals (mainly GA mice) being bred rising as a proportion of all studies; this accounts for 43% of all procedures (in 2021). Other common uses included basic research (51%), regulatory purposes (21%), translational/applied research (27%) and for other purposes (1%).
Broadly speaking, basic research involves investigation into understanding key mechanisms via which something occurs or functions. The most common areas focused upon in basic research were the nervous system (23%), immune system (20%) and oncology (12%). In contrast, applied research attempts to address diseases through prevention and development of treatments. The most common areas of research were animal diseases and disorders (48%), human cancer (15%), and human infectious disorders (12%). Finally, regulatory research is carried out to satisfy the legal requirements necessary to enable materials, products, and devices to be licensed for use. Regulatory procedures are usually carried out during the final stages of research and development and focuses on safety and efficacy. The most common procedure in 2021 was toxicity and other safety testing (52%).
“This year there has been a large increase in the proportion of applied research which has increased from 10% in 2020 to 27% in 2021. Whilst there was a large decrease in the proportion of regulatory procedures, which has decreased from 33% in 2020 to 21% in 2021. The rest of the purposes for experimental procedures have remained relatively stable since 2014.”
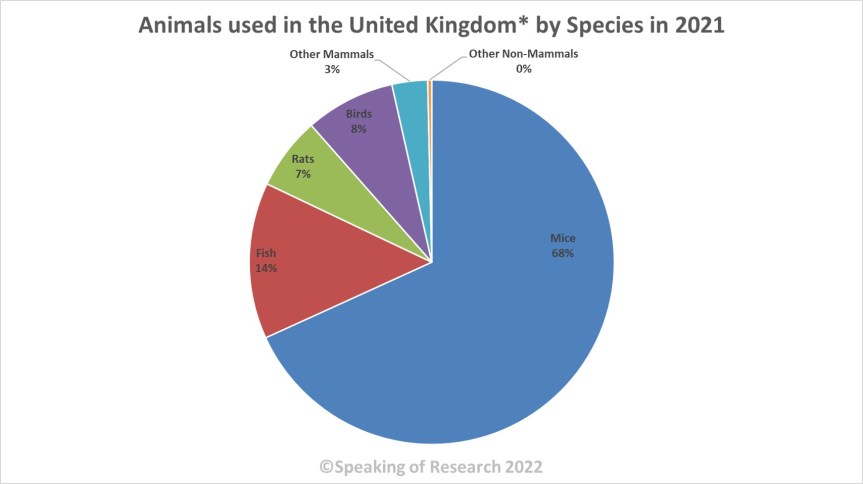
Looking at the species used we can see, mice, rats, birds and fish account for over 96% of all procedures on animals in Great Britain. Dogs and cats account for 0.14% and primates account for around 0.09%. Invertebrates such as fruit flies and nematode worms are widely used by researchers but are not covered by the Animals (Scientific Procedures) Act and are thus not included. The only invertebrate included in the statistics are cephalopods, though none have been used in recent years.

The UK statistics include retrospective reporting of suffering. Rather than just submitting license proposals to the Home Office that include estimated levels of suffering, the researchers now have to report on what was actually seen (using a variety of measures). Unfortunately, the statistics put these in two separate tables (Table 3.1 and 8). We have combined them to get severity for all procedures in 2021 – see definitions on page 23 or click here). It shows that 83.7% of procedures are sub-threshold, non-recovery or mild, and involved minimal pain or suffering for the animals. 13% of studies were moderate and 3% were severe.

~Speaking of Research
