This Guest Post is by Stuart Peirson, Associate Professor in the Nuffield Laboratory of Ophthalmology and chair of the 3Rs sub-committee at The University of Oxford. This article was originally posted on the website of the National Centre for the Replacement, Refinement & Reduction of Animals in Research (NC3Rs) and is reprinted with full permission. This article explains how Oxford is supporting the 3Rs, please read out page on UK research regulations and the 3Rs for more information.
 The University of Oxford is one of the world’s leading centres for biomedical research, with outstanding strengths in both basic science and its clinical application. The University’s Policy on the Use of Animals in Scientific Research outlines the University’s commitment to ensuring that all those involved in animal-based research are proactive in pursuing the 3Rs, engage fully in the ethical review process, and fulfil their moral and legal responsibilities for the care and welfare of animals.
The University of Oxford is one of the world’s leading centres for biomedical research, with outstanding strengths in both basic science and its clinical application. The University’s Policy on the Use of Animals in Scientific Research outlines the University’s commitment to ensuring that all those involved in animal-based research are proactive in pursuing the 3Rs, engage fully in the ethical review process, and fulfil their moral and legal responsibilities for the care and welfare of animals.
Ethical review
Reflecting the enormous breadth of research across Oxford, the University currently holds over a hundred different project licences, with over a thousand personal licence holders. This poses a number of challenges for the coordination of ethical review as well as the dissemination of best-practice and advances in the 3Rs.
The critical element in this process is the Animal Welfare and Ethical Review Board (AWERB). All applications for project licences require the ethical approval of the University before they are passed to the Home Office. At Oxford, this involves a rigorous and objective process of ethical review that challenges scientists to justify their use of animals, and that requires them, where the use of animals is unavoidable, to minimise animal numbers and maximise animal welfare.
At Oxford the AWERB process consists of two principal elements. Firstly, a central Committee on Animal Care and Ethical Review (ACER) is responsible for setting policy, as well as reviewing applications involving the use of non-human primates, severe protocols or novel techniques. Secondly, Oxford also relies upon a network of Local Ethical Review Panels (LERPs), which consider all other project licence applications. All project licences are required to provide a written retrospective review for their LERP at two years and four years, providing a critical opportunity for the LERP to assess how project licences have applied the 3Rs in their research.
The 3Rs sub-committee
In addition to the ethical review process, the University also has a 3Rs sub-committee reporting directly to ACER, which receives copies of all retrospective reviews to identify key developments in the 3Rs across the University. These developments are combined to form a termly 3Rs newsletter, which also contains information on relevant workshops, lectures and courses, such as NC3Rs notifications.
In addition, the committee also recognises the achievements of particular groups in the application of the 3Rs, providing letters of commendation to those project licence holders who show particular commitment and dedication to the 3Rs.
Since the introduction of the University’s Policy on the Use of Animals in Scientific Research, all departments involved in such research are also required to have termly Departmental Animal Welfare meetings. These are attended by project and personal licence holders, vets, Named Animal Care and Welfare Officers (NACWOs) and animal care staff, and provide a valuable forum for discussion of advances in the 3Rs.
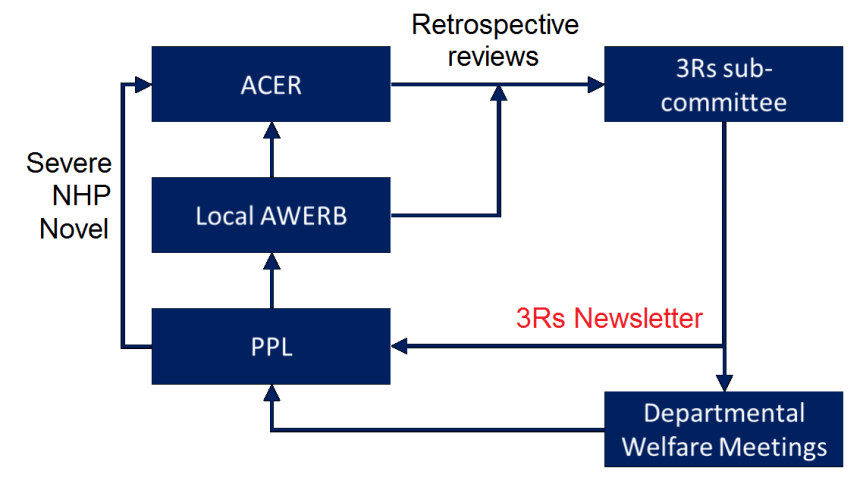
Summary of the role of the 3Rs sub-committee within the ethical review process
The 3Rs sub-committee also arranges lectures and workshops in areas it has identified as being important. For example, in 2013 we held a workshop on ‘Developments in Transgenic Mouse Models’, involving speakers from both Oxford and MRC Harwell, covering subjects ranging from colony management and background strains to existing transgenic resources and developing new transgenic models.
Working together
Biomedical Services (BMS) is an independent University Department of the Medical Sciences Division. BMS provides world class animal facilities that provide accommodation and care for its animals, delivered by professionally trained staff. A central principle of the University’s policy is the commitment to a culture of care, encouraging a team approach to animal work that fosters good communication and collaboration between all those working with animals in scientific research.
To facilitate this, in addition to their role on AWERBs, BMS staff, (including vets and NACWOs), routinely attend Departmental Welfare meetings, providing an informal opportunity for project and personal licence holders to discuss their work. The regular interaction has encouraged BMS staff and academic scientists to work together to achieve both high quality research and animal welfare.
Finally, BMS also provides key central services, such as the University’s new online training and competency records and colony management systems. Furthermore, practical veterinary assistance is also provided, such as a recent series of workshops on aseptic technique.
The future
Whilst Oxford has made great progress in implementation of the 3Rs throughout its scientific research programme, more can still be done. For example, we are currently building a ‘3Rs Knowledge Bank’ containing key and up-to-date references and protocols relating to best practice in the 3Rs.
We are also currently working on a University Strategy for the 3Rs, based upon the NC3Rs publication ‘Implementing an Institutional Framework for the 3Rs’. This will ensure that the 3Rs are thoroughly embedded in the research activities of the University, and that when animal research is necessary, it is conducted to the very highest of standards.
Professor Stuart Peirson
