This guest post is by Lisa Krugner-Higby, DVM, PhD. Dr. Krugner-Higby is a scientist and also a research veterinarian within the Research Animal Resource Center at the University of Wisconsin-Madison. Dr. Krugner-Higby’s research is in development of extended-release formulations of analgesic and antimicrobial drugs. She previously worked in anti-HIV drug development.
I am always fascinated by the idea promoted by some animal rights activists – repeated in many versions and for many decades – that all preclinical biomedical research can be conducted using in vitro cell culture. I have never found one of them who has spent much time working with cell culture. On the other hand, I have spent approximately seven years of my life working with cell cultures, looking at the stainless steel back wall of a laminar flow work station day after day. One thing I can say about immortalized cell lines, or cells that reproduce indefinitely, is that they are not alive in the same way that a mouse is alive.

The first thing that a graduate student learns when they begin to work with cell culture is how to take cells that have overgrown the sterile plastic flask they inhabit and put them into a fresh flask with fresh growth medium. It’s called ‘splitting’ the number of cells and ‘passaging’ them into a new home. Split and passage, split and passage… I knew that with every passage, the cell line became a little more different than normal cells and even a little more different than the original cell line. The remedy for this type of genetic drift was to freeze low passage cells in liquid nitrogen and re-order the line from the repository when the low passage stocks were depleted. I was careful with my sterile technique, cleaned the laminar flow hood, and used a new sterile pipet every time in order to avoid contamination of my cells. Unfortunately, the day came when I opened the incubator door and the flasks were black and fuzzy with fungus, and all of my carefully tended cells were dead. An anguished conversation with the tissue culture core technician revealed that this happened every Spring in North Carolina when the physical plant turned on the air conditioning for the year, blowing a Winter’s worth of fungal spores out of the ductwork and into the air. She recommended doing other things for about 6 weeks until the spore load had blown out of the ducts. I have had other cell line disasters in my scientific career: the malfunctioning incubator thermostat that turned a colleague’s two months’ worth of cell culture growth into flasks of overheated goo or that generally reputable vendor that sold us a case of tissue culture flasks that were not properly sterilized resulting in clouds of bacteria in the warm, moist, nutrient-rich environment of the incubator.
I never thought to ask, in those early days, if the cells that I fussed, worried, and wept over, were actually the cells that they were supposed to be. Raji Cells, A549s, U937s, I knew them all, worked with them every day, and never doubted that they were the cells that I thought that they were. I knew that some cell lines had been contaminated with the HeLa cell line. HeLa cells are very hardy and could spread quite easily from one flask to another. But I thought that was in the past. It turns out that there was more to the story than I realized. Cell lines have a provenance, like paintings or other works of art. They have an origin, a laboratory where the line was first isolated and propagated. From there, it may have been distributed to other laboratories and to repositories like the American Type Culture Collection or ATCC. Some cell lines are used by only a few laboratories, and some become used very widely and in a large number of biomedical disciplines. Whereas some paintings are intentionally forged, many cell lines have now been shown to be unintentionally forged. A recent article in the journal Science estimated that 20% of all immortalized cell lines are not what they were thought to be1.
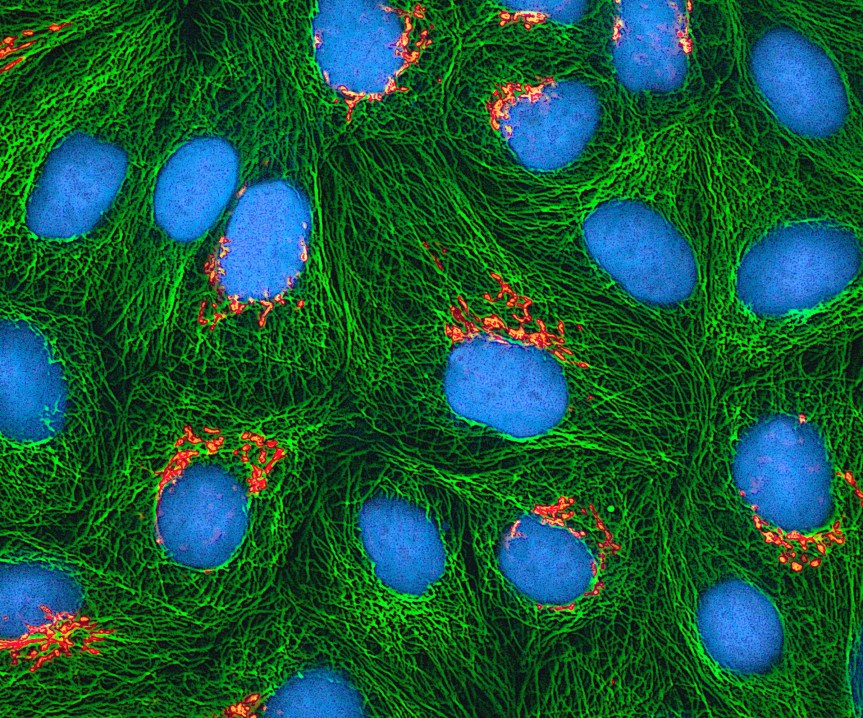
Multiphoton fluorescence image of cultured HeLa cells with a fluorescent protein targeted to the Golgi apparatus (orange), microtubules (green) and counterstained for DNA (cyan). Nikon RTS2000MP custom laser scanning microscope. National Institutes of Health (NIH).
We now have better methods of identifying cell lines by their DNA, called short tandem repeat (STR) profiling, and scientific journals are beginning to require this testing for cell lines prior to publication. Currently, 28 scientific journals require STR profiling to establish cell line provenance prior to publishing a manuscript from a particular laboratory. Some scientists are also beginning to create catalogs of contaminated cell lines in an attempt to quantitate the damage done by some misidentified, but widely studied, cell lines. The same Science article, notes that the International Cell Line Authentication Committee (ICLAC) maintains a database of misidentified cell lines that now numbers 475 different lines. A cell line geneticist, Dr. Christopher Korch, recently estimated that just two of the immortalized cell lines that were found to be misidentified, HEp-2 and INT 407, have generated 5,789 and 1,336 articles in scientific journals, respectively. These studies cost an estimated $713 million dollars and generated an estimated $3.5 billion in subsequent work based on those papers1. This is because the usual trajectory for testing a new therapeutic modality, especially in cancer research, is to test a compound or technique in cell culture. It will then be tested in mice that express a tumor derived from the cultured cancer cells. If those studies are successful, the compound and/or technique undergoes further toxicity testing in other animal models before entering its first Phase I trial in human volunteers.
A lot of compounds that show early promise in cell culture and in cell line-injected mice turn out not to have efficacy in animal models or in human patients. Sometimes this is simply a matter of the compound being too toxic to organs or cell types that are not represented in the initial cell culture. Often, the reason why particular compounds or strategies fail is not known, and most granting agencies are not keen to fund laboratories to find out why things don’t work. I have wondered if the failure of some compounds or techniques is in part due to misidentified cell lines. I have also wondered if it is a reason why testing in animal models, particularly in animal models with spontaneously-occurring tumors, is necessary.
Testing anti-cancer compounds in models of spontaneously-occurring tumors in animals and/or testing in human tumor cells taken directly from patients and injected into mice (the ‘mouse hospital’ approach) is more time and resource intensive than screening in immortalized tumor cell lines. This approach, however, has the advantage of knowing that the investigator is not just treating misidentified HeLa cells in error. It is always necessary to go from in vitro cell culture models to in vivo animal models to confirm the viability of a therapy.
Science makes claim to no enduring wisdom, except of its method. Scientists only strive to be more right about something than we were yesterday, and efforts are underway to weed out misidentified cell lines. But the fundamental issues behind cell line misidentification highlight one of the reasons why we should not rely on immortalized cell lines without animal models, and why granting agencies should fund more studies to try to identify the disconnect between the results of in vitro and in vivo studies when things do not go as planned. That is a part of good science and part of creating better cell culture models to refine, reduce, and sometimes replace the use of animals in biomedical research.
Lisa Krugner-Higby, DVM, PhD
1) Line of Attack. Science. 2015. Vol. 347, pp. 938-940.
