March 8th 2021
Renée Hartig, PhD
Researchers at the University of Wisconsin-Madison recently published a scientific report detailing an innovative procedure to treat one of the most prevalent neurodegenerative diseases amongst the older population: Parkinson’s disease. This procedure utilized grafts of brain cells to replenish dopamine neurons in the adult brain. Grafts, along with stem cell therapy, have the potential to help restore damaged tissue to a healthy state. However, this treatment option is still experimental and has been proven complicated in humans. For one, we know that if the body’s immune system detects new tissue as foreign, there can be attacks on the new cells and their survival within existing tissue is jeopardized. Methodological validation and proof-of-concept studies are vital to this sector of research in order to bring such potential to the mainstream and human patients. Thus, researchers worked with a model for Parkinson’s disease in ten rhesus monkeys to study the success of brain cell grafts in mediating Parkinsonian symptoms.
Parkinson’s disease, which is characterized by an impairment of dopamine transmission in the brain, can result in dyskinesia, motor, emotional and cognitive impairments. At present, there is no cure for Parkinson’s disease, but there are a few available treatments to improve the lives of those suffering from its unwanted and, often, devastating effects. Upon diagnosis and onset of symptoms, such as dyskinesia, hand tremors and walking gait disturbances, one pharmaceutical option (L-DOPA) has been shown to express efficacy in human patients for up to an order of many years. However, for some patients, long-term L-DOPA use can lead to negative side effects, for instance, hyperkinesia (or involuntary hyper-extensive movements), where the influence of supplemental dopamine seems to become less effective.

Aside from this widely used pharmaceutical treatment option, surgical interventions occupy a niche in Parkinson’s disease treatment. One method already in place involves the implantation of an electrode stimulator in the brain to mimic the effects of lost dopamine neurons—to supply the brain with energy and electrical pulses. With this intervention, neurosurgeons specifically target where it seems the brain could most use the external input. Of course, it is not a fail-safe option, and its surgical nature can make it just as technically challenging as grafting brain cells.
Nevertheless, brain cell grafting provides an additional treatment for Parkinson’s disease and may provide a greater capacity for tissue restoration. In the recent study, published in the journal Nature Medicine, induced pluripotent stem cells (iPSCs) were derived from different individuals, cultured, and re-engineered in vitro to become brain cells, specifically dopamine neurons. These neurons were injected into the basal ganglia, or region of the brain involved in relaying dopamine signaling to parts of the striatum and other areas of the cortex. When successfully transplanted, these dopamine neurons can revitalize the underlying population of cells and form new connections where others had been previously lost.
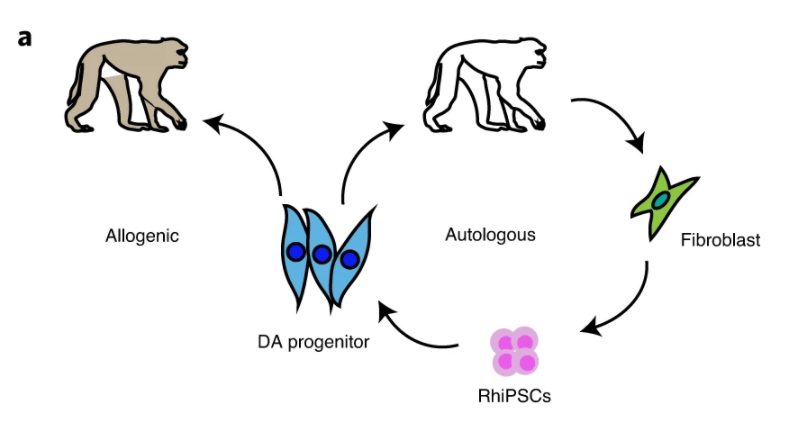
This study by Tao and colleagues examined, in a monkey model of Parkinson’s, how self-derived dopamine cells (autologous) and dopamine cells derived from another monkey (allogenic) impacted the successful integration of these new cells into the existing brain network and, therefore, the effectiveness of the procedure as a Parkinson’s therapy. Already within 6-12 months, improvement in Parkinsonian symptoms, such as fine-motor skill impairment, was significantly better when the graft was made from self-derived cells. In contrast, grafts derived from a different monkey did not improve dopamine connectivity nor were Parkinsonian symptoms reversed in this case. This indicated that brain cell grafts stand a better chance of success when they are from self-derived cells. The new findings validate a protocol for self-derived brain cells and their potential for successful integration in brain tissue that can inform planning for similar interventions in humans.
Given the gravity of any invasive procedure, which includes tissue extraction and transplantation, a refined protocol for operation is directly related to intervention success. The information gathered from this research will aid clinical utilization of self-derived brain cells and justify a procedure, which can undoubtedly be more labor-intensive, where at the end of the day each cell graft would have to be individually tailored without much room for streamlining a one-fits-all process for every patient. However, knowing that the work and medical intervention would yield a higher chance of success and substantially improve quality of life makes the procedure and efforts worthwhile. For further information on the role of animal research in the development and optimization of treatment options for Parkinson’s disease, refer to this recent post on how Progress in Parkinson’s disease depends on Primate and other #AnimalResearch.
