Welcome to this week’s Research Roundup. These Friday posts aim to inform our readers about the many stories that relate to animal research each week. Do you have an animal research story we should include in next week’s Research Roundup? You can send it to us via our Facebook page or through the contact form on the website.
- CRISPR gene editing technique argued to be probably safe. In a previous research roundup, we highlighted the ongoing debate with respect to the safety of the CRISPR-Cas 9 gene editing technique. In that study, it was argued that despite using this technology to restore sight to mice, when looking at the whole genome of the animals, hundreds of areas other than that targeted DNA sections were affected in three of those mice. In a new preprint of an article (non-peer reviewed), researchers argue that the differences that were found were simply a consequence of genetic relatedness rather than unexpected mutations. While it is still far from certain which side is correct, such dialogue and debate highlight the stringency that most treatments that eventually make it humans go through before introduction to the general population — much in part because of animal research.
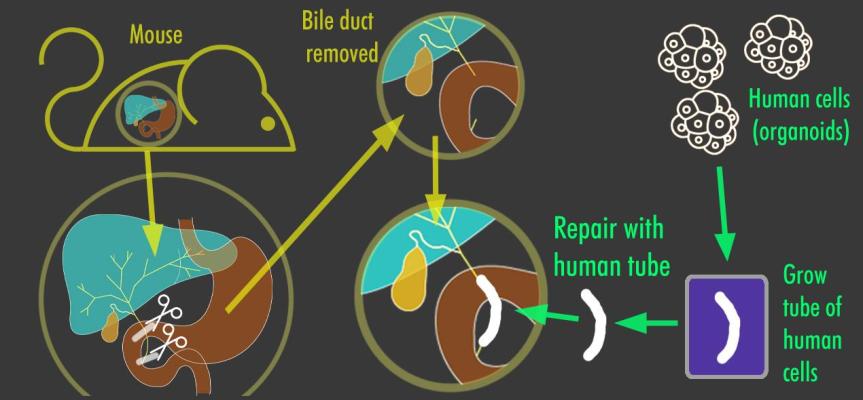
- Artificial bile ducts successfully grown in the lab and transplanted into mice could help treat liver disease in children. Scientists in Cambridge, UK, have developed a new method for growing artificial bile ducts in the lab and successfully transplanting them into lab mice — a development that could one day be used to treat liver disease in children. The discovery could also reduce the need for liver transplants in these patients. The researchers extracted healthy cells (called cholangiocytes) from bile ducts and grew them into functioning 3D structures known as biliary organoids. Researchers then grew the organoids on a “biodegradable collagen scaffold” in order to shape the organoid into a tube, which they then transplanted into mice to replace damaged bile ducts. The transplants were successful, and the animals survived without any further complications. The scientists emphasized the “power of tissue engineering and regenerative medicine that these results demonstrate. “These artificial bile ducts will not only be useful for transplanting, but could also be used to model other diseases of the bile duct and potentially develop new drug treatments,” said Dr. Fotrio Sampaziotis, lead author on the study.
The research was published in Nature Medicine.
- Antibodies of mother halt placental transmission of cytomegalovirus in monkeys. In our research roundup two weeks ago, we highlighted the debilitating effects of CMV on infants. Approximately 50% of all humans over the age of 40 harbour the CMV, and over 1 million infants a year are infected worldwide. Here, for the first time, researchers studied whether the offspring of mothers exposed to CMV specific antibodies, would confer protection to their offspring from the virus in utero. In the first experiment they found that dosing with CMV antibodies prevented abortion of the fetus and in a second experiment found that a higher dose completely blocked transmission of the virus.While the virus in rhesus macaques is not identical to that in humans (RhCMV) much can be learned from studying this derivative of the disease in non-human animals – similar to the study of SIDS in our understanding of AIDS. In terms of the applicability of this work, lead author, Cody Nelson, a PhD student at DUke university says “Ending congenital CMV infection is likely going to require an effective vaccine given before pregnancy, similar to how the rubella vaccine has eradicated congenital rubella syndrome in the Americas.” This research was published in the journal JCI Insights.
- Thermal imagery of bat hibernation suggests group behavior for combating white-nose syndrome. Insect-eating bats play a large role in pest control services, likely saving the U.S. agriculture industry upwards of $3 billion a year. However, white-nose syndrome is a fungal disease that has been spreading rapidly across North America for the past decade and is causing steep declines in bat populations. A recent study by researchers at Massey University in New Zealand, used temperature-sensing cameras on hibernating bats with white-nose syndrome to better understand how some bats survive white-nose syndrome during hibernation, while others do not. Interestingly, they found that a species of bat (Indiana bats, Myotis sodalis) — that is less affected by the disease than others (little brown bats, M. lucifugus)– slowly warmed up as a synchronous group, which may have enabled body temperatures to be less conducive to fungal growth and increase the bat’s ability to survive the disease. Not only does this basic research help us towards finding solutions to mitigate the declining bat population, but it also may help scientists in the future to combat disease in astronauts who will hibernate during long-term space travel.

- The 2017 winners of the Universities Federation for Animal Welfare (UFAW) Medal for Oustanding Contributions to Animal Welfare Science were Professor Sandra Edwards and Professor Jeff Rushen. The medal celebrates exceptional achievements in animal welfare over a number of years. Sandra Edwards, Professor of Agriculture at Newcastle University, works on improving welfare in pigs through improvements in housing, environmental enrichment, health monitoring and more. Professor Jeff Rushen an Adjunct Professor at the University of British Columbia, works on cattle welfare, stereotypic behaviors in animals, and looking at how to reduce fear in animal-human relationships.
- Cilene Lino de Oliveira has won the Basel Declaration Society 2017 Award for Education in Animal Research. Oliveira, from the Department of Physiological Sciences at the University of Santa Catarina, teaches the University’s “Laboratory Animal Care and Welfare” course. The award will give her the opportunity to do an EU course in animal welfare at the Institute for Laboratory Animal Science at the University of Zurich.
