July 7, 2019
Recent campaigns targeting nonhuman animal research in the US have illustrated how strategies and groups that are old, new, and convergences of the two can affect science and the public that benefits from new knowledge and advances in science and medicine. We’ve written about a number of these issues over the years, including a post last week about research animal retirement. Today we take a broad look at some current campaigns, some of the mechanisms for various types of legislative efforts, and some suggestions for action.
What’s in the news?
For months, scientific research at federal agencies has been the target of a range of campaigns whose core goal is to end studies that involve nonhuman animals. The studies are publicly funded because they are aimed at advancing scientific knowledge, medical treatments, and challenges to public health.
The work is conducted by federal agencies with familiar names and long histories of accomplishments. They are agencies charged with ensuring public health and using science to evaluate risks from foods, drugs, pollutants, infectious and other diseases and to address those threats with evidence-based practices, policies, and treatments. While those public agencies have long served societal and global needs, they have also found themselves under attack. In the case of scientific research that depends on studies of nonhuman animals, those attacks have recently included campaigns against:
- FDA: Research at the Food and Drug Administration (FDA) to better understand how nicotine and the chemicals that often co-occur with its consumption affect adolescent health;
- VA: Research at the Veterans Administration (VA) that has advanced treatments for spinal cord injury and other serious health threats that affect our nation’s veterans and others;
- USDA: Research at the Department of Agriculture (USDA) aimed at public safety from toxoplasmosis;
- NIH: Research at the intramural branch of the National Institutes of Health (NIH), with a specific focus not on any particular topic of research or health, but rather on any studies that involve nonhuman primates.
- NIH: Research funded by the NIH that uses rats, mice, and other nonhuman animals in a broad range of studies aimed at advancing scientific knowledge to benefit public health.
A mix of strategies have been employed in campaigns against this research. Among them are the wide range of now (very) familiar approaches: website and social media posts, petitions, letters and op-eds, press conferences and press releases, billboards, event disruptions, and various other methods of garnering attention.
Legislative efforts
As notable are the steady stream of bills introduced by members of the US Congress. The examples below are bills introduced in Congress over the past six months. In most cases, the bills aim at a single type of animal (dogs, cats, primates). In all cases, the bills aim at federally funded research. The accompanying media releases typically express little about concern for the same kind of animals in privately funded, commercial research. We’ll come back to this point, but first, a quick review:
- PUPPERS– Preventing Unkind and Painful Procedures and Experiments on Respected Species. Reps. Dina Titus (D-NV), Brian Mast (D-FL); reintroduced Feb 2019.
- KITTENS– Kittens in Traumatic Testing Ends Now Act. Sen. Jeff Merkley (D-OR); reintroduced March 2019.
- Primates– Primate Protection and Research Modernization Act. Sen. Cory Booker (D-NJ); introduced 2018, not reintroduced in 2019.
- HEARTS– Humane and Existing Alternatives in Research and Testing Sciences Act. Rep. Lucille Roybal-Allard (D-CA); introduced Valentine’s Day 2019.
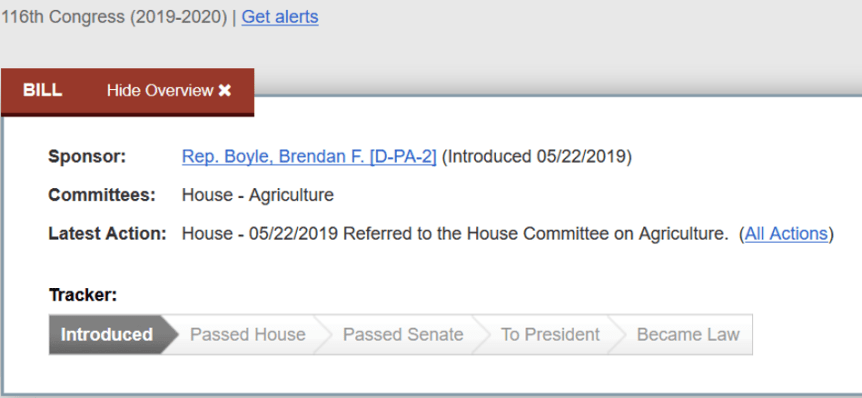
- AFTER– Animal Freedom from Testing, Experimentation and Research (AFTER) Act. (Reps. Jackie Walorski (R-Ind.) and Brendan Boyle (D-Pa.); introduced May 22, 2019.
In recognition of SR’s international readership (and some fun facts about civics), we’ll start out with a quick basic primer on the US legislative process. First, all of the above are bills—proposed legislation that has been introduced by one or more members of Congress, in either the Senate or the House of Representatives. After a bill is introduced it is typically referred to a relevant committee for consideration. It must then go through a process including a vote in the chamber where the bill was introduced, a vote on a companion bill in the other chamber, and reconciliation of any differences in the two bills, before going to the US President for signature or veto. A great deal of legislation is proposed by members of Congress—over 18,000 pieces in 2016-17 and 2017-18 and over 5,000 so far in 2019-20. A much smaller number make it through to a vote by one of the chambers, less to a vote by both chambers, and even less to a positive vote by both chambers making it the law, after it is signed off by the president.
How can constituents and others learn about proposed legislation?
Proposed legislation is a first step that merits careful attention and consideration in a democratic society. One way to learn more about legislation that has been introduced is to read the bill. Bills can be read in their entirety on the congressional website that provides the text, actions taken on the bill, a list of its sponsors and cosponsors, and other relevant information. For example, The HEARTS Act can be found here: https://www.congress.gov/bill/116th-congress/house-bill/1209
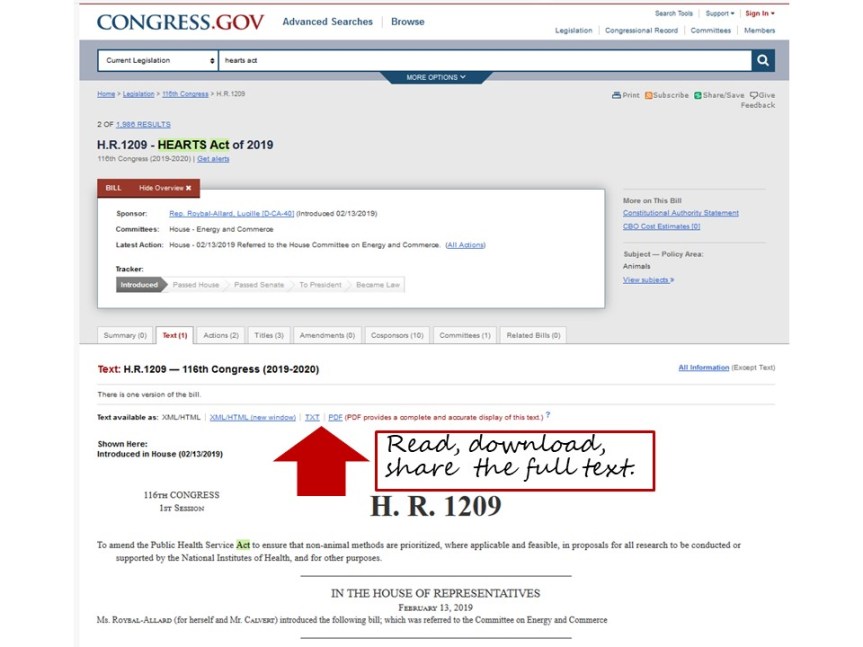
Who supports the legislation?
If you’re wondering whether the bill has broad support, or whether your congressional representatives have supported it, you can also find that information on the same page that shows the bill, text, and actions. For instance, for the HEARTS Act, the page provides a list of 11 current sponsors. The AFTER Act, introduced by Reps. Brendan Boyle (D-PA) and Jackie Walorski (R-IN) on May 22 in the House, has 14 co-sponsors. Co-sponsors for HEARTS and AFTER include many members of the congressional animal protection caucus, whose history, goals, and members can be found here. The caucus includes 130 of the 535 representatives and senators that together comprise the US Congress. In a recent Hill blog article, caucus leaders Reps. Vern Buchanan (R-FL) and Earl Blumenauer (D-OR) predicted 2019-20 to be a successful year for their legislation:
This session, the Congressional Animal Protection Caucus has an even more ambitious agenda.
With more than 130 members of Congress, our caucus is committed to passing legislation that combats animal cruelty, ends painful experimentation on animals and permanently bans the slaughter of American horses for human consumption” (emphasis added).
The congressmen then go on to highlight six bills, none of which include any reference to “ending” animal research or testing, but instead aim at animals used for food, entertainment, sport, or show.
What is the status of the bill?
The same congress.gov page shows what actions have been taken, providing the public with information about who introduced the bill, when it was introduced, whether and when it was referred to a congressional committee for further consideration, what committee it was referred to, whether it has been voted on, and so on. The “Tracker” graphic illustrated below shows each of the steps: Introduced, Passed House, Passed Senate, To President, and, finally, Became Law.
Why read the original source?

There are many ways to learn about proposed legislation. In many cases, the congress member who introduces the bill will also issue a media release and highlight key provisions of the bill on their own website or through media (for examples, see here, here, here).
At the same time, advocacy groups, lobbyists, political parties, and others will publicize the proposed legislation. But each of those will be a version with its own emphasis. What is written in the proposed legislation is what congress members may vote on. Moreover, what is written in the proposed legislation is often what requires critical evaluation and can benefit from additional information, rebuttal, or elaboration.
For instance, in the HEARTS Act, many scientists familiar with the NIH process for review of scientific proposals and the multiple levels of oversight on animal research, might be surprised at the rationale put forward for the proposed legislation. In fact, the legislation duplicates many procedures and policies already in place, including consideration and use of alternatives to nonhuman animals. Further, much of the associated media coverage fails to provide a serious consideration of the role nonhuman animal studies play in basic research that is at the heart of NIH’s ability to deliver scientific advances that benefit humans and other animals.
Reality check
Legislation that is unlikely to advance is proposed for many reasons. It fails to advance for many more. It is probably safe to say that members of Congress are unlikely to propose legislation that they believe would be unpopular with their constituents or those whose favorable opinion Congress members value. If few speak up and little attention is given to potentially broad negative consequences of proposed legislation, it may seem that there is relatively little downside to supporting it. While apparently obvious, adopting such a lens to scan the proposed legislation listed above can provide a useful guide to the scientific community and others who value serious consideration of public health.
A basic political reality can be that it is relatively easy to make a case about “saving” kittens, puppies, and nonhuman primates. By contrast, it is more complex to present the moral dilemma and societal benefits of scientific research that depends on ethical, responsible use of nonhuman animals in research. The question then is how to ensure that public decisions are informed, accounting for the breadth and long-term range of likely consequences of those decisions. Providing accurate information about humane treatment of nonhuman animals is one piece of this and has been a focus for outreach and public communication. But ultimately, this approach may not do much to dissuade legislators from proposals like those highlighted here.
What is needed instead is a clear articulation of the downside, the negative consequences, the harms—for the public, for society, the environment, other animals—of ending, jeopardizing, or compromising research. In other words, for one example: the use of a small number of dogs in research at the VA has resulted in discoveries and treatments that can help veterans and others with spinal cord injury (read more here and here, for example).

Ending that research has a consequence. It is likely to harm the available treatments for those with spinal cord injury.
Proposed legislation that advances may also fail for reasons that are practical and fiscal. In some cases, a bill that may seem desirable also has to be balanced with its cost were it to be enacted. Before signing on, congressional representatives may seek an estimate of that cost. The analysis can come from the Congressional Budget Office (CBO). Under the US legislative system, that analysis is also made public and can be found at the same site as the legislation.
For an example of a CBO analysis on animal research relevant legislation, we can look to the 2000 Chimpanzee Health Improvement, Maintenance, and Protection Act (CHIMP Act; CBO analysis here). Or to the Great Ape Protection and Cost Savings Act of 2011 that failed to pass (CBO analysis here). CBO analyses, when requested and completed, are posted along with the bill. What the analysis provides is a public summary of what the bill will cost if enacted—arguably, one (of many) necessary pieces of information for public decision-making.

Bill-adjacent legislative efforts
Another form of legislative pressure is found in what is called “report language” that can accompany a bill emerging from a congressional committee (or subcommittee). Report language is not a bill or proposal for a law, but rather text that can be inserted into a report that accompanies a bill. Any member of the originating committee or subcommittee can insert language into the report, unless another member objects, or the language contradicts or neutralizes the original language. The impetus for report language may be via external stakeholders—including advocacy, scientific, political, or other organizations—who draft report language to further their own agenda. The stakeholders work with congressional offices with which they have forged strong relationships to get the language inserted into the report.
Unlike legislation, report language is non-binding and does not mandate a response or action. Given that report language is non-binding, it has little to no impact on how the legislation is enacted or enforced by the agency that is the subject of the bill’s provisions, except if the agency leadership interpret the language as the will of the congressional committee and decide to act on it. This happened in 2015, for example, when the report accompanying the House appropriations bill that covers the NIH (FY2016 LHHS) included language requesting NIH to “conduct a review of its ethical policies and processes with respect to nonhuman primate research subjects….” (H. Rept 114-195, pp 73; image below).

In response to this nonbinding request NIH convened a workshop in September 2016. The announcement and conclusions of the workshop provoked media coverage and conversation aptly summarized by the Science magazine headline “ Workshop on ethics of monkey research earns cheers and boos” (pdf of full report here).
2019 Report Language Aimed at Nonhuman Primate Research at Intramural NIH Program
A recent example of report language used to target research with nonhuman animal research include a move by Rep. Roybal-Allard (D-CA), who inserted language about NIH’s intramural nonhuman primate research program in a report accompanying the Fiscal Year 2020 House Appropriations Bill for Labor, Health and Human Services, and Education and Related Agencies (LHHS; see page 114). The paragraph (see graphic below) “urges the NIH to accelerate efforts to reduce and replace the use of nonhuman primates with alternative research models” and directs the agency to prepare a report that addresses six items. The six include an overview and summary of NIH projects that use nonhuman primates; an explanation and review of alternatives to the use of nonhuman primates for research, areas including where alternatives are not available; “a detailed strategy and timeline for the reduction and replacement of NIH primate research with alternative research methods;” and a standard procedure for retirement of animals no longer needed in research.
The language was widely reported as though it were a mandate to reduce NIH’s nonhuman primate research. A headline from the science magazine Nature, for instance, read:
US lawmakers propose plan to reduce primate research at National Institutes of Health: Spending bill would require the agency to identify alternatives to research with monkeys and other non-human primates.
Meanwhile, a group opposed to federal research with nonhuman animals headlined a blogpost about the report language with “Progress: Funding panel directs NIH to cut primate testing.” Each of these headlines illustrates how report language can be represented as far more, or something different, than it is.
Who is leading?
In some cases, the campaigns are led, or promoted, by groups, like PETA, that are on record in opposition to the use of animals by humans for any purpose—food, clothing, research, entertainment. As we’ve written before, these groups may be absolutists, opposed to the use of nonhuman animals regardless of the purpose, whether there are alternatives to achieve the same goal, and regardless of the harm that would result (see here, here, or here for discussion of foundational arguments and absolutism). In other cases, the campaigns are led by groups that are less clear in their position, claiming not to be opposed to animal use, only to federal agencies doing so. For instance, White Coat Waste, a conservative group we’ve written about previously (see here, here, here), has been closely involved in many of the recent campaigns. Many other groups, including scientific societies, are often involved in advocating for various legislation that affects human use of nonhuman animals, here we’ve focused primarily on recent campaigns generally opposed to animal research.
What’s the problem?
Legislative efforts to advance public health, meet societal needs, protect nonhuman animals and the environment are all commendable. However, these efforts should be based in evidence and fact, and not grounded in ideology and spurious interpretations of data. Critical consideration must be given to what is proposed to be implemented and how.
For example, efforts to protect nonhuman animals can fall into the different realms in which they are used by humans, including for entertainment, agriculture, conservation, research, and so on. One can make reasonable arguments for and against the use of animals in each of these domains as well as for their protection. Credible information, such as what standards and systems of oversight for the use of animals in each domain should be transparently reported and evaluated to provide a sound basis for critical consideration and decision-making. The use of nonhuman animals for research, for instance, has multiple systems and levels of oversight already in place which address the balance of the potential harms of not doing such research or of conducting the work, and the potential benefits of doing so. At the same time, this does not imply that existing systems cannot be improved–there is often room for continuing improvement in any human system. What we’d argue is that it is in the public’s interest that such changes be grounded in empirical evidence rather than anecdote and that defined metrics of benefit associated with the change are specified clearly.
Summary: What can you do?
Legislative maneuverings to impede, hinder, and ultimately abolish all research with nonhuman animals are not in the best interest of humans, other animals, the environment, and society writ large. They threaten sound public policy that can have serious ramifications for scientific advancement, medical progress, and public health.
We’ve provided a quick primer on the mechanics of some recent efforts aimed at impacting animal research through legislative endeavors. For some of our readers these campaigns will be familiar, but the question will remain: What can I do about it? For non-scientists, one thing to do is to be informed, ask questions, share factual information, and contact your legislators to let them know that you expect policy to be evidence-based, with serious consideration of the full range of consequences for decisions.
As an informed scientific citizen, the obligation of a research community member to respond to these challenges is not actually that daunting. Most major scientific organizations constantly monitor federal legislative and regulatory actions and keep their membership informed with relevant details. So, the next time you get an email about the introduction or reintroduction of a piece of legislation that is detrimental to important research with nonhuman animals, you could:
- Contact your university’s government relations office to inquire about how they intend to respond to the bill that would seriously hinder vital research that is being conducted on your campus. If you don’t already know what office that is, or the people in it, take a few minutes to ask around– who in your university or company works on legislative issues? Are they already working on issues that affect scientific research with animals? Consider asking whether there is anything you can do to help, whether they have materials about animal research on your campus, or at your company, whether they will update you on how they advocate for research.
- Write a thoughtful, fact-based message to your own Congressional representatives informing them about the importance of animal research in general and the relevance of the particular research, or animal model under attack. Explain the potential consequences of the legislation. Urge them not to support that and similar legislation. Don’t start or stop there though. Provide factual, concise, and accessible information that makes your case and helps others understand not just why animal research is conducted, but also how animals are cared for, how humane treatment is ensured, how the existing regulation, multi-tiered systems for review and oversight work. Welcome them to contact you if they have questions or would like more information.
- Contact your scientific societies or professional associations and ask them how they are responding to legislation and other efforts to impede scientific research. Ask for specifics and ask how you can help lend your voice and expertise.
- Spread the word among colleagues, urging them to similar actions.
These are just a few of the many ways that you can take an active role to contribute to public dialogue that informs policy decisions–as many scientists, scientific, and advocacy organizations already do. We’ll be back with future posts focused on analysis of specific pieces of current legislation and legislation-adjacent efforts that could affect scientific research with nonhuman animals. In the meantime, let us know how you, your university, or your scientific organization are helping to advance thoughtful, serious, and evidence-based approaches to policy that affects scientific research with nonhuman animals.
Speaking of Research


