May 20th 2021
Agnès Lacreuse, PhD
Our world is aging at an unprecedented rate. The number of older people is projected to double every 20 years, and reach about 17% of the total population by 2050. These figures are alarming, as age is the primary risk factor for developing devastating diseases such as Alzheimer’s disease (AD). In the US, 1 in 9 people over 65 currently have AD, maybe someone you know, or a loved one, is one of these people. The number will continue to increase, with 13 million people expected to have AD by 2050. Animal research has resulted in many discoveries that help us to understand the disease and its biological mechanisms (for instance, how and why the hallmarks of the disease, the amyloid plaques and tau tangles, interact and progress from one brain region to another). A first step to progress in curing or treating a disease is understanding it well enough to improve early diagnosis and to develop better treatments. Next, the promising treatments must be selected and evaluated. In the case of diseases of the brain, like AD, studies in rodents provide important basic biological information, but may not yield therapies that are successful in humans. Indeed, to understand why the human brain is so vulnerable to AD and to find effective treatments, we need to study the brain and behavior of other primates.
Our laboratory is dedicated to scientific studies that can advance knowledge and treatment of diseases like AD. We study a nonhuman primate called the common marmoset, a small monkey (300-500 g) about the size of a rat. The marmoset is a highly valuable model to study aging processes because it has a short lifespan of about 10 years and their cognitive function declines with age. To study a human across their lifespan might take 60-90 years. Thus, we can study the marmoset brain and behavior as they age within this shorter time window and try to understand why some animals age well, while others show a significant decline in intellect and brain function.

Our studies are focused on cognitive function (thought and intellect). The monkeys are trained to voluntarily perform a variety of cognitive tasks on a touchscreen computer to obtain one of their favorite rewards, a banana milkshake. We also make the most of the animals by evaluating their motor and emotional abilities, their sleep, behavior, and physiology to have a complete picture of their aging characteristics. We also study their brain, using non-invasive neuroimaging like that which is done with humans, and we study brain tissue for disease markers postmortem because it is more difficult to do so in humans who have died from AD. From this and other research, we know that older marmosets develop some of the hallmarks of AD in their brain.
Why does the research on aging and AD in marmosets matter? By relating their neuropathology to their behavior we can start to understand the mechanisms underlying the aging process, detect predictors of age-related decline and potentially detect AD earlier (yielding a better diagnosis) or develop new therapeutics against AD (yielding better treatments) to slow down or stop this debilitating neurodegenerative disease in its tracks.
Women’s Health
Another important aspect of our research is focused on to women’s health, because 2 out of 3 cases of AD are cisgender women, for reasons that urgently need to be understood. In addition, women with AD typically experience worse symptoms than men. We do not yet understand these sex differences, but they are vastly important, because men and women may need to be given different types of treatments to prevent and alleviate symptoms. Sex differences are difficult to study in humans, because of many factors that interact with sex, such as gender, race, education, lifestyle, medication, exercise, etc. By studying animals like the marmoset, scientists can ask direct questions about the role of sex differences while considering these other interacting factors separately.
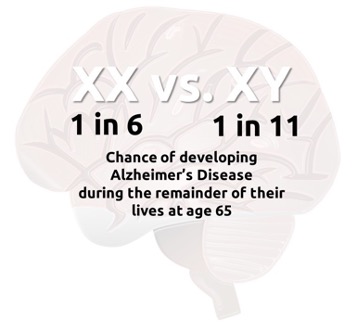
Currently, scientists suspect that sex chromosomes and the sex-specific hormones that they control may exacerbate AD symptoms. But to understand this fully requires primate models like marmosets. Thus, our laboratory is studying how changes in sex hormones, especially estrogens, affect brain and cognitive aging in female marmosets to better understand sex differences in AD and contribute to future sex-specific therapies.
Our work in marmosets aims to develop a new treatment that would safely alleviate these severe side effects and help breast cancer survivors live a better life.
~Agnès Lacreuse, PhD
Why does this research on how estrogens affect AD symptoms in female marmosets matter? The decrease of estrogens that occurs at menopause in previously cycling women is associated with a number of adverse effects on the brain, cognition, metabolism and many other systems. A majority of women will experience cognitive deficits, sleep problems and hot flashes. These are serious symptoms and they should not be mocked and dismissed with a stereotypical “they will get through it” response. It is crucial to understand individual differences in symptom severity and how these symptoms interact with one another, because for some middle-aged women, the menopause transition will be a pivotal point that will determine their future aging trajectory — including the risk of developing AD. This work also has important implications for women with breast cancer. Breast cancer patients are often given treatments that completely block estrogens and that give rise to a number of side effects that are debilitating. Our work in marmosets aims to develop a new treatment that would safely alleviate these severe side effects and help breast cancer survivors live a better life.
Why does the National Institutes of Health fund our research?
Like much of the basic, pre-clinical research in the biomedical and behavioral sciences, the studies highlighted above are funded by the National Institutes of Health (NIH). What that means is that NIH recognizes the translational value of our work and its potential for public health. The NIH has a long history of funding basic science because it is the foundation for discoveries that lead to the understanding of human health and disease, and, in turn, new ways to prevent and treat disease, reduce human suffering, and improve public health. A testament to the value of investing in basic research can be found in the fact that NIH-funded research has contributed to 96 Nobel Prizes in Physiology and Medicine (full list here), contributing to breakthroughs in antiviral drugs for hepatitis C, cancer therapy, stem-cell therapy, and improving vaccine effectiveness—to name a few since 2010. Notably, the NIH budget for 2021 of $42.9 billion is just around 3% of the massive $1.4 trillion spending bill approved by Congress back in December.
Many NIH-funded studies depend on research animals, including not only monkeys, but also rats, mice, pigs, and other species. You can read more about these at this fact page. For all of these studies, ethical justification and research approval is required before any research can begin. Proposals for research undergo a rigorous review process by multiple different groups of experts who examine the scientific question, methods, procedures, and animals’ treatment. The deciding factors for funding and approval of the research include everything from the scientific review and NIH review that critically evaluates the public health relevance and potential knowledge to be gained, to the federally-mandated committees that closely monitor the animals’ treatment. Throughout the time that the studies are conducted external oversight continues through both institutional committees and federal agencies. You can read more about how studies move from a scientist’s idea here and more about approval and oversight here. Like other basic research that relies on studies of nonhuman animals, our laboratory engages in and supports humane and transparent animal research. To learn even more about what we do, please check our web site at https://www.lacreuselab.com/.
AD is a public health crisis that needs urgent solutions. I am personally convinced that studies in nonhuman primates, including the marmoset, are key to better understand the causes and mechanisms underlying the disease and discover effective therapeutics.

